The goal here is simple: the patient should get the same therapeutic effect whether they take the brand-name drug or the generic version. This isn't just a suggestion; it's a legal requirement born from the 1984 Hatch-Waxman Amendments. Essentially, the FDA wants to see that there is no significant difference in how the active ingredient becomes available at the site of action when you take the same dose under the same conditions.
The Core Requirements: Pharmaceutical vs. Bioequivalence
Before a company even starts a clinical trial, they have to nail two distinct markers: pharmaceutical equivalence and bioequivalence. You can't have one without the other.
Pharmaceutical equivalence is the "blueprint" phase. The generic must have the same active ingredient, the same dosage form (like a tablet or a capsule), the same strength, and the same route of administration. If the brand-name drug is a 50mg extended-release tablet taken orally, the generic must also be a 50mg extended-release tablet taken orally.
Bioequivalence is where the actual science happens. It's the proof that the drug actually behaves the same way inside a human body. The FDA focuses on the Reference Listed Drug (or RLD), which is the brand-name version. The manufacturer must show that their test product is "bioequivalent" to this RLD, meaning it reaches the target area of the body in a way that produces the same clinical result.
Decoding the 80/125 Rule
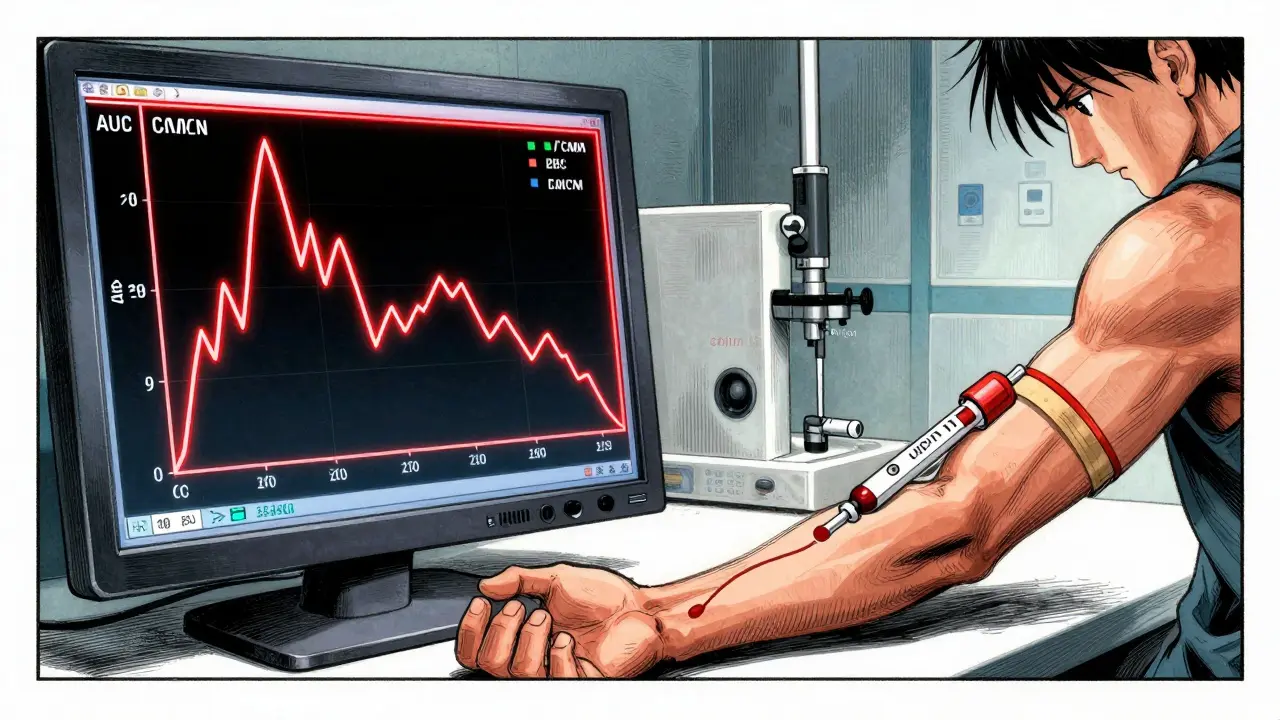
How does the FDA actually measure "equivalence"? They don't just look at a few patients and call it a day. They use rigorous pharmacokinetic (PK) studies that track how the drug moves through the body. There are two primary metrics they watch: Cmax (the maximum concentration of the drug in the plasma) and AUC (the Area Under the Curve, which represents the total drug exposure over time).
To pass, manufacturers must follow the 80/125 rule. This means the 90% confidence interval for the ratio of the geometric means between the generic and the RLD must fall between 80% and 125%. If your drug's absorption is too fast or too slow-even if it's only slightly outside this window-the FDA will likely reject the application.
| Metric | What it Measures | FDA Standard (90% CI) | Significance |
|---|---|---|---|
| Cmax | Peak Plasma Concentration | 80% to 125% | Ensures the drug doesn't hit a toxic peak or fail to reach therapeutic levels. |
| AUC | Total Drug Exposure (Absorption) | 80% to 125% | Ensures the body absorbs the total intended amount of the drug. |
How These Studies Are Actually Conducted
Most systemic drugs require in vivo studies. This means testing the drug in living humans. Typically, the FDA requires trials involving 24 to 36 healthy volunteers. These participants are usually tested under fasting conditions to get a clean baseline of how the drug is absorbed. In some cases, the FDA will also demand "fed" studies to see if eating a meal changes how the drug works.
These trials are expensive and high-stakes. A single study can cost anywhere from $500,000 to $2 million. Because the data is so critical, the FDA requires strict adherence to Good Laboratory Practice (or GLP) regulations. This covers everything from how samples are stored to how the lab equipment is calibrated. One mistake in documentation can lead to a Complete Response Letter (CRL), which is essentially a "no" from the FDA.
When You Can Skip the Human Trials: Biowaivers
Not every drug needs a full-scale human trial. For certain products, the FDA allows a "biowaiver," which lets manufacturers prove equivalence through lab tests instead of human subjects. This is a huge win for companies, as it can shave 6 to 12 months off the development timeline.
Biowaivers are typically granted using the Q1-Q2-Q3 framework:
- Q1 (Qualitative): The generic has the exact same active and inactive ingredients.
- Q2 (Quantitative): The amount of those ingredients and the concentration are identical.
- Q3 (Physicochemical): The pH, particle size, and other physical properties are the same.
This approach is common for parenteral solutions (injectables) or ophthalmic drops. For topical creams, the FDA might accept In Vitro Release Testing (IVRT) and In Vitro Permeation Testing (IVPT) to show the drug penetrates the skin at the same rate as the RLD.
Special Cases: Narrow Therapeutic Index (NTI) Drugs
The 80/125 rule works for most drugs, but some are too dangerous for that kind of leeway. These are called Narrow Therapeutic Index (or NTI) drugs. For an NTI drug, a tiny change in concentration can be the difference between a cure and a toxic overdose.
Think of drugs like warfarin (a blood thinner) or levothyroxine (for thyroid issues). For these, the FDA tightens the screws. Instead of 80-125%, they often require a much stricter acceptance range, such as 90-111%. If you're manufacturing an NTI drug, your margin for error is almost zero.
Common Pitfalls and How to Avoid Them
The failure rate for the first cycle of ANDA approvals is surprisingly high, often around 43%. Why? Most of the time, it's not because the drug doesn't work, but because the bioequivalence study was botched. Common mistakes include using a sample size that's too small to be statistically significant or failing to follow the Product-Specific Guidances (PSGs).
The FDA has published over 2,100 of these PSGs. They are essentially "cheat sheets" that tell you exactly how to design your study for a specific drug. Companies that follow these guides see a massive jump in success rates-roughly 68% for first-cycle approval compared to just 29% for those who wing it.
The Future of Bioequivalence: Complex Generics
As we move toward 2026 and beyond, the FDA is focusing more on "complex generics." These are drugs like inhalers, long-acting injectables, or drug-device combinations where the delivery mechanism is just as important as the chemical itself. For these, the FDA is starting to use Physiologically Based Pharmacokinetic (or PBPK) modeling. This uses computer simulations to predict how a drug will behave in a human body, potentially reducing the need for massive clinical trials.
What is the difference between bioavailability and bioequivalence?
Bioavailability refers to the rate and extent to which a single drug is absorbed and becomes available at the site of action. Bioequivalence is a comparison between two different drug products (usually a generic and a brand name) to see if they have the same bioavailability.
Can a drug be pharmaceutically equivalent but not bioequivalent?
Yes. A drug can have the same active ingredient, dose, and form (pharmaceutically equivalent), but if the inactive ingredients (excipients) or the manufacturing process change how the drug dissolves in the stomach, it may not reach the bloodstream at the same rate, making it not bioequivalent.
Why does the FDA use a 90% confidence interval instead of a simple average?
A simple average can be skewed by one or two outliers. A 90% confidence interval ensures that the result is statistically robust and that the drug's performance is consistent across a wide range of patients, not just a lucky few in a small sample.
What happens if a bioequivalence study fails?
If the study fails the 80/125 rule, the manufacturer must reformulate the drug-changing the fillers, binders, or coating-and run the study again. If they cannot achieve bioequivalence, they cannot market the drug as a generic substitute for the RLD.
How long does it typically take to get FDA approval after bioequivalence is proven?
While it varied wildly in the past, the current timeline from ANDA submission to approval typically ranges from 14 to 18 months, provided there are no major deficiencies in the bioequivalence data.

